ABCSG 62 / CAMBRIA-2 Overview
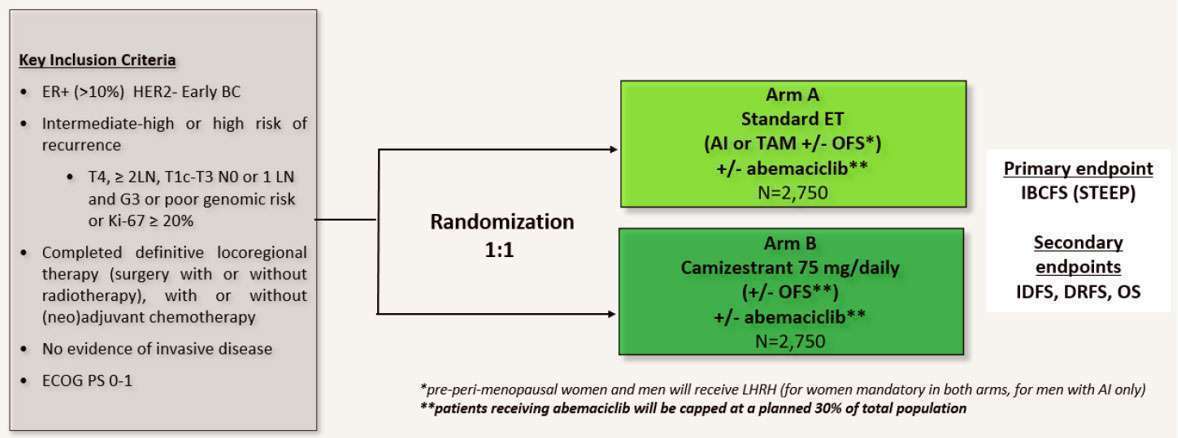
A Phase III, Open-Label, Randomised Study to Assess the Efficacy and Safety of Camizestrant (AZD9833, a Next Generation, Oral Selective Estrogen Receptor Degrader) Versus Standard Endocrine Therapy (Aromatase Inhibitor or Tamoxifen) as Adjuvant Treatment for Patients with ER+/HER2- Early Breast Cancer and an Intermediate-High or High Risk of Recurrence Who Have Completed Definitive Locoregional Treatment and Have No Evidence of Disease.
| Start: | (global): 10/2023; FPI 10/2023 (national): 04/2024 |
| Coordinating Investigator AT: | Daniel Egle, Innsbruck |
| Sample: | 5500 (global), 128 (national) |
| Study design: (Click to enlarge) |
 |
Share on

