ABCSG 53 / TAXIS / SAKK 23/16 Study Details *
A multicenter randomized phase III trial to compare tailored axillary surgery with or without axillary lymph node dissection followed by radiotherapy in patients with clinically node-positive breast cancer.
| Study Start: | 10/2019 |
| Recruitment open until: | 12/2024 |
| Coordinating Investigator: | Fitzal, Florian; Vienna |
| Sample Size: | AT: 100 Global 1.500 |
| Study Design: (Click to enlarge) |
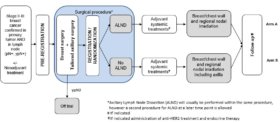 |
Treatment
Patients after TAS are randomized to undergo the following:
-
Arm A
ALND then regional nodal irradiation excluding the dissected axilla as a target volume.
-
Arm B
Regional nodal irradiation including the full axilla.
All patients undergo adjuvant whole-breast irradiation after breast conserving surgery and chest wall irradiation after mastectomy.
Primary Objective
To show that TAS and axillary radiotherapy (RT) is non-inferior to axillary lymph node dissection (ALND) in terms of disease-free survival of breast cancer patients with positive nodes at first presentation in the era of effective systemic therapy and extended regional nodal irradiation.
Endpoints
Primary endpoint
-
Disease-free survival (DFS)
Secondary endpoints
-
Quality of Life (QoL)
-
Overall survival (OS)
-
Breast cancer-specific survival (BCSS)
-
Time to local recurrence (TTLR)
-
Time to regional recurrence (TTRR)
-
Time to distant recurrence (TTDR)
-
Reported morbidity outcomes: lymphedema and decreased
-
range of shoulder motion
-
Adverse events
-
Late radiotherapy-related adverse events
-
Surgical site infections (SSI)
Inclusion Criteria
Inclusion criteria at pre-registration
-
Written informed consent according to ICH/GCP regulations prior to any trial specific procedures.
-
Breast cancer, node positive detected by palpation or imaging (with or without planned neoadjuvant treatment).
-
Female or male aged ≥ 18 years.
-
Ability to complete the Quality of Life questionnaires.
Inclusion criteria at registration
-
Node-positive breast cancer (histologically or cytologically proven both in primary tumor and in lymph node) AJCC/UICC stage II-III (all molecular subtypes allowed):
-
Node-positivity detected by imaging and non-palpable (iN+) and confirmed by pathology.
-
Node-positivity palpable (cN1-3) and confirmed by pathology.
-
Occult breast cancer is allowed, if biopsy-proven axillary lymphatic metastasis is present.
-
-
Eligible for primary ALND or SLN procedure with frozen section and either:
-
Newly diagnosed.
-
Isolated in-breast recurrence or second ipsilateral breast cancer after previous breast conserving surgery and sentinel procedure and at least 3 years disease free and no prior axillary dissection or axillary RT.
-
-
Most suspicious axillary lymph node clipped.
-
Baseline Quality of Life questionnaire has been completed.
-
WHO performance status 0-2 .
-
Adequate condition for general anesthesia, breast cancer surgery and radiotherapy.
-
Women of child-bearing potential are using effective contraception, are not pregnant or lactating and agree not to become pregnant during trial treatment and thereafter during the time recommended by the guidelines for adjuvant systemic therapies. A negative pregnancy test before registration is required for all women of child-bearing potential.
Men agree not to father a child during trial treatment and thereafter during 6 months.
Inclusion criteria at randomization (intraoperatively)
-
Node-positive breast cancer (histologically or cytologically proven both in primary tumor and in lymph node) AJCC/UICC stage II-III (all´molecular subtypes allowed):
-
Node-positivity initially detected by imaging and non-palpable and residual disease confirmed by pathology (including residual ITCs) in SLN or non SLN in case of prior neoadjuvant treatment.
-
Node-positivity initially palpable and residual disease confirmed by pathology (including residual ITCs) in case of prior neoadjuvant treatment.
-
Exclusion Criteria
Exclusion criteria at pre-registration
-
Stage IV breast cancer.
-
Clinical N3c breast cancer (clinical N3a and clinical N3b are allowed).
-
Clinical N2b breast cancer (clinical N2a is allowed).
-
Contralateral breast cancer within 3 years.
-
Prior axillary surgery (except prior sentinel node procedure in case of in-breast recurrence).
-
Prior regional radiotherapy.
-
History of hematologic or primary solid tumor malignancy, unless in remission for at least 3 years from pre-registration with the exception of adequately treated cervical carcinoma in situ or localized non-melanoma skin cancer.
-
Treatment with any experimental drug within 30 days of preregistration.
-
Any Other serious underlying medical, psychiatric, psychological, familial or geographical condition, which in the judgment of the investigator may interfere with the planned staging, treatment and follow-up, affect patient compliance or place the patient at high risk from treatment-related complications.
Exclusion criteria at randomization (intraoperatively)
-
Absence of clip in the specimen radiography.
-
Palpable disease left behind in the axilla after TAS.
-
No SLN identified in the axilla.
Share on